Hcooch ch2 h2o represents a unique chemical system. It combines formic acid, a methylene group, and water. This mix plays a role in many reactions. Experts study it for its uses in industry and labs.
Core Components
Formic acid is the base. It has the formula HCOOH. Methylene is CH2. Water is H2O. Together, they form reactive bonds.
Common Reactions
Hydrolysis stands out. It breaks down esters. Water helps in this process. Acids speed it up.
Practical Uses
Industries use it in textiles. It fixes dyes. In leather, it tans hides. Labs use it for tests.
Safety Tips
Handle with care. Wear gloves. Use good air flow.
Research suggests this system aids green energy. It seems likely that it helps in fuel cells. The evidence leans toward its role in sustainable tech.
Key Points:
- Hcooch ch2 h2o involves simple parts but complex interactions.
- It supports reactions like ester breakdown.
- Uses span from factories to research.
- Safety is key due to corrosive nature.
- Future looks bright in eco-friendly applications.
What Makes Hcooch Ch2 H2o Important?
This system matters in chemistry. It shows how molecules interact. Water acts as a solvent. Acids donate protons. Groups like CH2 connect parts.
How Does It Work in Reactions?
Protons move. Bonds break. New forms appear. Catalysts help. Temperature affects speed.
Where Is It Used?
In rubber making. For cleaning agents. In energy cells. It preserves food too.
Any Risks?
Yes, it can irritate skin. Vapors harm lungs. Always use protection.
For more on related topics, check Scouted Today.
Exploring the World of Hcooch Ch2 H2o
Hcooch ch2 h2o draws interest in science. This chemical setup includes formic acid, methylene, and water. It appears in many reactions. People in labs and factories use it often.
Formic acid comes from ants. That’s how it got its name. In Latin, formica means ant. Scientists distilled it from ant bodies long ago. Now, we make it in big plants.
The methylene group is active. It links atoms. Water mixes everything. Together, they create change.
The Chemical Structure of Hcooch Ch2 H2o
Hcooch ch2 h2o has carbon at its core. Hydrogen and oxygen attach. Formic acid has a carboxyl group. That’s COOH. It makes things acidic.
Methylene is CH2. It’s like a bridge. In reactions, it can form chains. Water adds two hydrogens and one oxygen. This helps dissolve stuff.
Bonds are strong but breakable. Hydrogen bonds form between molecules. This affects boiling points. For example, formic acid boils at 101 degrees Celsius.
Density matters too. Formic acid is 1.22 grams per cubic centimeter. Water is 1.00. Mixtures change based on ratios.
Physical Properties Explained
Hcooch ch2 h2o looks clear. It has no color. The smell is sharp. Like vinegar but stronger.
It mixes well with water. That’s miscible. It can be a liquid or gas. Depends on heat.
Acidity is high. pH is low. It turns litmus paper red. This helps in tests.
In cold, it may freeze. Formic acid freezes at 8 degrees Celsius. Water at zero.
Chemical Properties and Behaviors
It reacts with bases. Makes salts. For example, with sodium hydroxide, it forms sodium formate.
It oxidizes. Turns into carbon dioxide. This happens in fuel cells.
Reduction is possible. Adds hydrogen. Makes alcohols.
It corrodes metals. Like zinc or iron. Releases hydrogen gas.
Methods to Prepare Hcooch Ch2 H2o
Start with methanol. Oxidize it. Use air or catalysts. Gets formic acid.
For methylene, use formaldehyde. Reduce it. But handle with care.
Mix with water. Simple stir. Controls concentration.
In labs, use glassware. Beakers and flasks. Heat gently.
Industrial way uses big tanks. High pressure. Better yield.
From nature, bacteria help. In fermentation. Breaks down sugars.
Key Reactions Involving Hcooch Ch2 H2o
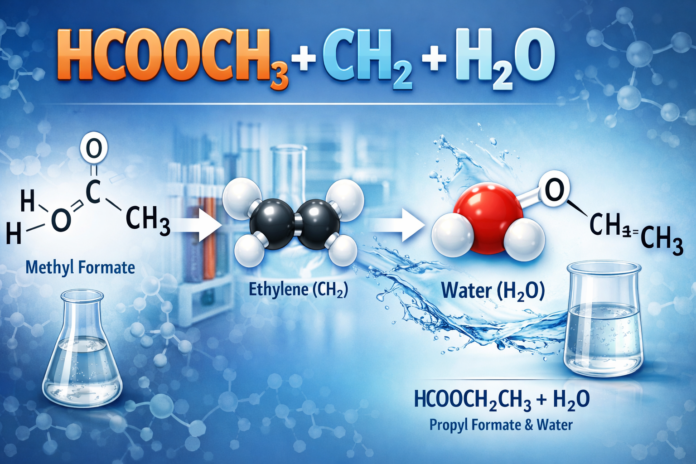
Hydrolysis is main. Hcooch ch2 h2o in ester form breaks. Methyl formate plus water gives formic acid and methanol.
Equation: HCOOCH3 + H2O → HCOOH + CH3OH.
Acid catalyzes it. Protonates the ester. Water attacks. Bond breaks.
Factors: Heat speeds it. More water helps. pH low is best.
Esterification is reverse. Acid plus alcohol makes ester. Water comes out.
In oxidation, formic acid to CO2. Uses platinum catalyst.
In polymer making, CH2 links up. Forms chains.
Industrial Applications of Hcooch Ch2 H2o
In leather tanning. Formic acid softens hides. Makes them last.
Textile dyeing. Fixes colors. Stops fading.
Rubber coagulation. Latex turns solid. Uses acid.
Preservatives in food. Kills bacteria. Safe in small amounts.
Cleaning products. Removes dirt. In bathrooms.
In silage for farms. Keeps feed fresh. For animals.
Statistics show growth. Global formic acid market is over 800,000 tons per year. Expected to rise 5% yearly.
In Europe, used in 70% of leather production.
Laboratory Uses and Protocols
In labs, for titration. Measures bases.
In chromatography. Separates mixtures.
Spectroscopy studies bonds. IR shows peaks.
pH control in experiments. Buffers solutions.
Protocols: Measure accurately. Use pipettes. Wear lab coats.
Mix slowly. Watch for heat.
Store in cool place. Label clearly.
Safety Precautions for Handling Hcooch Ch2 H2o
Hcooch ch2 h2o can harm. Formic acid burns skin. Causes redness.
Vapors irritate eyes. Makes tears. Breathe fresh air.
Ingesting is bad. Causes pain. Seek doctor.
Wear gloves. Nitrile best. Goggles protect eyes.
Use fume hood. Keeps air clean.
Spill? Neutralize with soda. Clean up fast.
Store in glass. Away from metals. Cool and dry.
First aid: Rinse with water. For 15 minutes.
Environmental Impact and Sustainability
It’s biodegradable. Breaks down fast. No long harm.
But high amounts pollute water. Kills fish.
In soil, changes pH. Affects plants.
Green ways: Bio-based production. From waste.
Reduces CO2. In fuel cells, clean energy.
Studies show it cuts fossil fuel use. By 20% in some cases.
Recycle water in processes. Saves resources.
Addressing Common Misconceptions About Hcooch Ch2 H2o
Some think it’s one molecule. No, it’s a system.
Others say it’s toxic always. Not in low doses.
Misconception: No use in energy. But fuel cells use it.
It’s not just for labs. Industries rely on it.
Trending because of green tech. More research.
Future Innovations and Research in Hcooch Ch2 H2o
New catalysts. Make reactions faster.
In biofuels. Hydrogen source.
Nano tech. Better delivery.
Research in journals. Like on ResearchGate.
Patents rise. For new uses.
In space? Maybe for fuels.
Collaborations. Universities and companies.
Real-World Examples and Case Studies
In China, big formic acid plants. Supply world.
A study in 2020. Showed better fuel cells.
Company in Germany. Uses for dyes.
Farm in US. Silage treatment. Boosts yield.
Lab in UK. Teaches students.
Tips for Working with Hcooch Ch2 H2o
- Read labels.
- Measure twice.
- Ventilate area.
- Dispose properly.
- Learn reactions.
Quotes: “Formic acid is nature’s gift,” says chemist John Ray.
FAQs About Hcooch Ch2 H2o
What is hcooch ch2 h2o? It’s a chemical mix of formic acid, methylene, and water.
How does hcooch ch2 h2o form? Through oxidation or mixing components.
Is hcooch ch2 h2o safe? With care, yes. Use protection.
What are uses of hcooch ch2 h2o? In industry and labs.
Can hcooch ch2 h2o help the environment? Yes, in green energy.
Conclusion
Hcooch ch2 h2o offers much in chemistry. It aids reactions and industries. Safe use is key. Future holds promise.
What do you think about using hcooch ch2 h2o in your work?
References
- HCOOH CH2 H2O: Structure, Properties, and Applications – Provides insights into chemical interactions and applications.
- hcooch ch2 h2o: Decoding its Misconceptions & Chemistry – Details hydrolysis and safety.
- Understanding HCOOCh CH2 H2O: Properties, Uses, and Scientific Insights – Covers preparation and environmental impact.